Causes, symptoms, diagnosis, therapy - and classification of frequency therapy
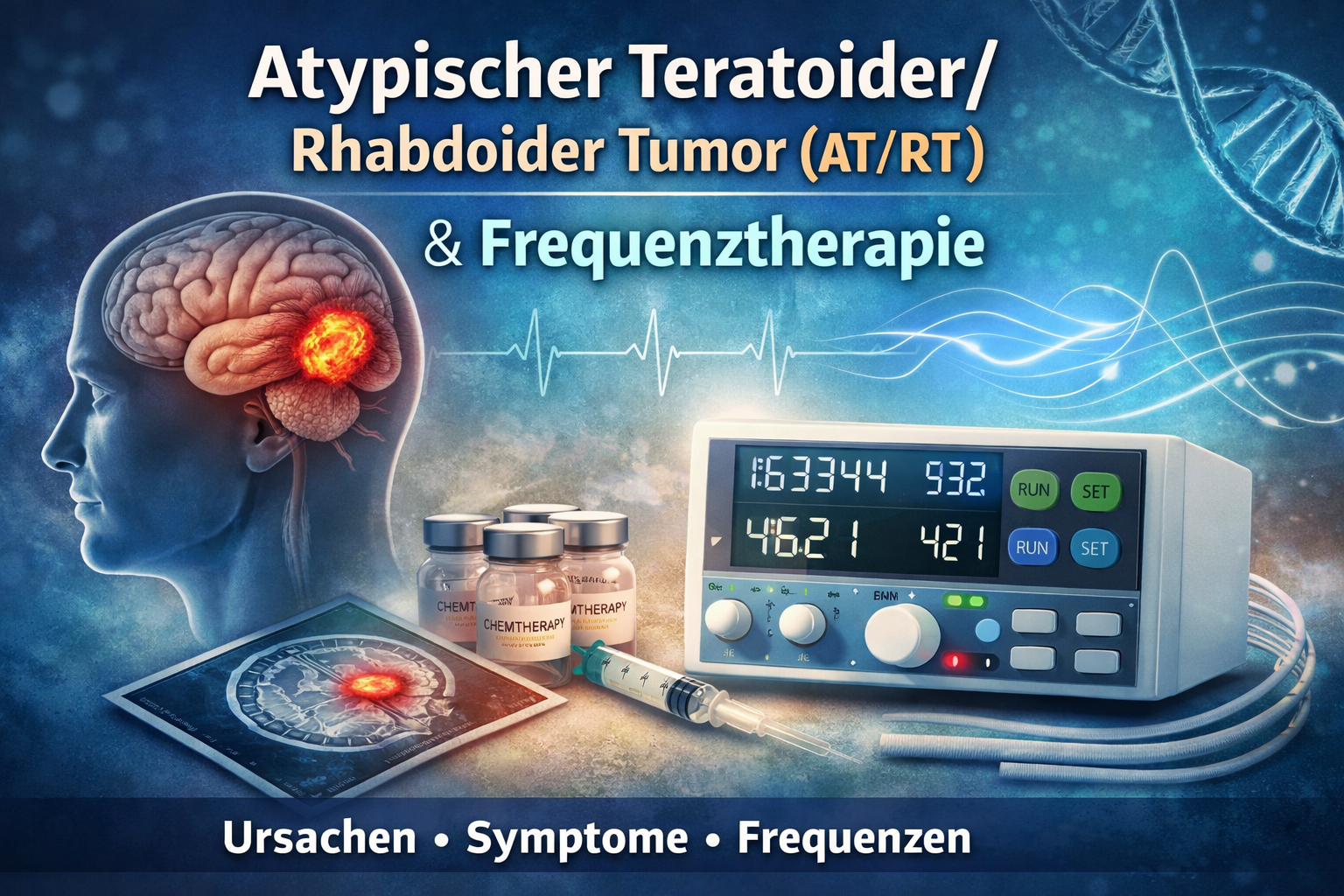
The Atypical teratoid/rhabdoid tumor (AT/RT) is a very rare, highly malignant (WHO grade 4) Tumor of the central nervous system (CNS), which predominantly for infants and small children occurs. AT/RT is one of the embryonal CNS tumors and requires a Rapid, specialized diagnostics and multimodal therapy in a pediatric oncology center.
What is an AT/RT?
AT/RT is one of the rhabdoid tumors; The most common site of rhabdoid tumors is the CNS. Characteristic are Genetic changes in the SWI/SNF chromatin remodeling complex, most frequently SMARCB1 (INI1); diagnostically, the Loss of INI1 expression in immunohistochemistry.
Epidemiology (rough classification):
- Very rare, but relatively more frequently in the 1st year of life than in later childhood years.
Symptoms
The complaints depend heavily on Localization and Tumor growth off. Common (non-specific) symptoms are
- Vomiting, nausea, Drinking/eating problems (in infants)
- Drowsiness, irritability, loss of performance
- Headache (for older children)
- Gait disturbances, coordination problems, visual disturbances
- Sign of a intracranial pressure (e.g. rapid deterioration)
Because AT/RT can grow rapidly, symptoms are often Rapidly progressive.
Diagnosis
Diagnostics are usually performed in specialized centers and typically include
- MRI of the brain and, if necessary, spinal cord (staging, spread)
- Neurosurgical biopsy or resection to confirm the diagnosis
- Neuropathology/Molecular Pathology (including INI1/SMARCB1 status)
- Depending on the situation: Cerebrospinal fluid examination (metastasis/dissemination)
The modern classification is based on the WHO classification of CNS tumors (CNS5).
Therapy in conventional medicine
The treatment is intensive and multimodal. Guidelines and major therapy protocols contain core components:
- Maximum safety operation (if possible extensive resection)
- Adjuvant chemotherapy (induction and consolidation phases)
- Radiotherapy (often focal; scope and timing adapted to age and risk)
A well-known example of an intensive protocol is COG ACNS0333 (surgery → induction chemotherapy → high-dose chemotherapy with stem cell rescue + radiation), which in publications reports survival improvements compared to historical controls.
Important: The treatment concept for very young children is weighed up particularly carefully (benefit/risk, late effects), often with highly specialized, age-adapted strategies.
Forecast
AT/RT is considered to be aggressive; The prognosis depends, among other things, on:
- Age at diagnosis
- Extent of the resection
- Metastasis/dissemination (M status)
- molecular subgroups/biological characteristics
- Therapy intensity and radiotherapy options
Research and new therapeutic approaches
As AT/RT is often combined with SMARCB1 loss is associated with the development of the disease, targeted strategies are being investigated. One example is the inhibition of EZH2 (e.g. Tazemetostat) in studies for SMARCB1/SMARCA4-deficient tumors or matching molecular profiles.
Frequency therapy for AT/RT (EDTFL / CAFL)
Note in advance: AT/RT is a Life-threatening cancer, which immediately in special pediatric oncology treatment. Frequency therapy can - if at all only accompanying and not as a substitute be understood.
Frequency lists (EDTFL or CAFL - depending on availability)
currently not available
Conclusion
AT/RT is a Rare, very aggressive pediatric brain tumor (WHO grade 4), where today a Combined treatment of surgery, intensive chemotherapy and (usually) radiotherapy is the basis. Guidelines and protocols such as ACNS0333 show that intensive, structured approaches can improve outcomes - but treatment remains complex and belongs in highly specialized centers.
Disclaimer
The Frequency therapy is not recognized by conventional medicine. It represents no replacement for diagnostics or treatment by trained doctors or Alternative practitioner and may be used in particular for serious illnesses such as Cancer diseases not be used instead of evidence-based therapies.
Author: NLS Informationsmedizin GmbH, Herbert Eder